THE 2ND LAW OF THERMODYNAMICS FOR CLIMATE
The Second Law of Thermodynamics correctly stated reads: "In a natural thermodynamic process the sum of the entropies of the interacting thermodynamic systems never decreases."
The word "thermodynamic" refers not only to processes involving heat (thermo) but also to ones involving motion (dynamics) such as the redistribution of air molecules due to gravity when a sealed horizontal cylinder is rotated to a vertical position. Such a redistribution (with greater density forming at the base and less density forming at the top) is an example of the Second Law acting an increasing entropy.
Whenever entropy is increasing there will be a reduction occurring in unbalanced energy potentials. In the above example there was initially more potential energy at the top than at the bottom. When all such unbalanced energy potentials have fully dissipated, we have the state of maximum entropy which we physicists call thermodynamic equilibrium. In the case of the cylinder, that state will have at all levels the same sum of mean molecular (potential energy + kinetic energy) and, since only kinetic energy affects the temperature, we have both a non-zero density gradient and a non-zero temperature gradient. Each such gradient is the sole result of gravity acting on individual molecules. Since pressure is proportional to the product of temperature and density (by the Ideal Gas Law) the pressure gradient is merely a corollary. Many scientists (like Nikolov and Zeller) think that high pressure causes high
temperatures. It doesn't because an increase in temperature requires an increase in kinetic energy, in other words, an input of energy from some external source.
The process described above occurs (especially in calm conditions at night) throughout the troposphere of every planet with an atmosphere. Hence both the density gradient and the temperature gradient are one and the same state of thermodynamic equilibrium. So entropy may be thought of as a cumulative measure of progress towards this state of thermodynamic equilibrium which, by the way, is different from thermal equilibrium because it does not necessarily exhibit the same temperature at all levels.
So this is the process which the brilliant physicist Josef Loschmidt first explained in the 1870's ahead of his time. There was clearly an indication that to form the temperature gradient there had to be some heat going from cooler to warmer regions. Scientists have been skeptical of this because they think (incorrectly) that the Second Law of Thermodynamics is only ever about heat going from hot to cold.
That is not always true in a force field.
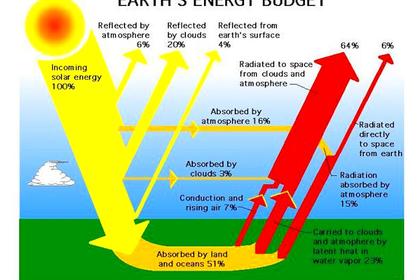
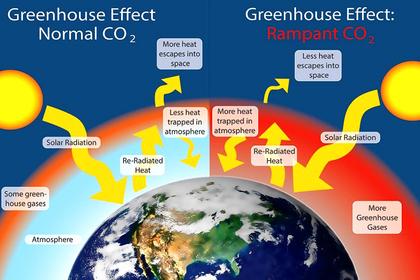
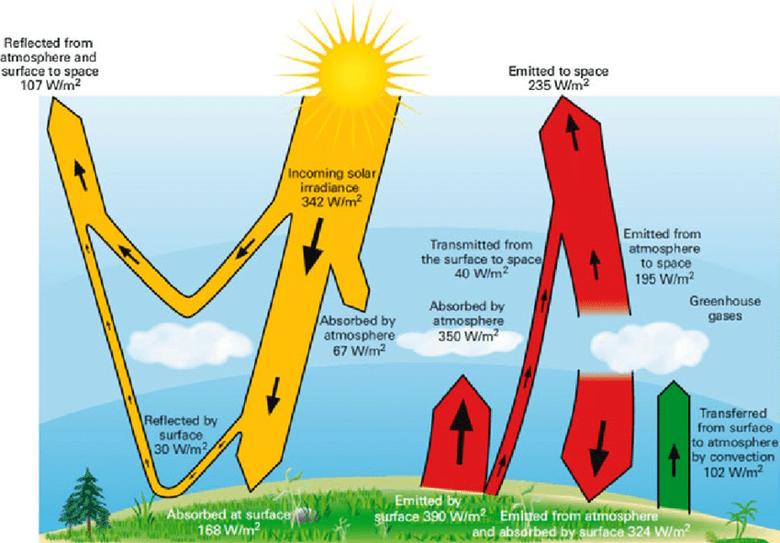
And that is what climatologists have completely overlooked. They have noted that the mean surface temperature is far hotter than the solar radiation to the surface, or even just to regions above in the troposphere, could explain in accord with the Stefan-Boltzmann Law. So they guessed (incorrectly) that the reason must have to do with the "back radiation" (as they called it) from so-called "greenhouse" gases (mostly water vapor) in the troposphere. So they added to the solar flux about twice as much radiation from the colder troposphere and used the net sum (less non-radiative cooling) in all their energy diagrams and computer modelling to quantify surface temperatures and any increases in such.
But the Stefan-Boltzmann Law only applies for a single source of radiation.
Climatologists realized that they were implying that radiation from cold regions in the troposphere was supposedly causing heat into the already-warmer surface. They made out that this was not a violation of the Second Law because, they thought, it could be excused seeing that there was more energy coming out of the surface by non-radiative and radiative processes. But the Second Law only ever applies to a single thermodynamic process or a set of interacting thermodynamic systems. The outward cooling, which may be in the afternoon rather than when the warming was occurring in the morning, is not in any way shape or form interacting with the inward "back" radiation.
Hence climatologists are wrong on three counts: (1) overlooking the Loschmidt gravito-thermal effect and (2) incorrectly applying the Stefan-Boltzmann Law calculations and (3) incorrectly excusing the assumed violation of the Second Law of Thermdynamics.
For those who may be asking what happens to the energy in the back radiation the answer is in my peerreviewed paper "Radiated Energy and the Second Law of Thermodynamics" published on several sites since 2012.
-----
This thought leadership article was originally shared with Energy Central's Energy Collective Group. The communities are a place where professionals in the power industry can share, learn and connect in a collaborative environment. Join the Energy Collective Group today and learn from others who work in the industry.
-----
Earlier: